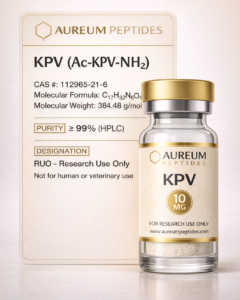
NAD+
Nicotinamide adenine dinucleotide, β-Nicotinamide adenine dinucleotide, Coenzyme I, Cozymase, DPN, Diphosphopyridine nucleotide, NAD, β-NAD

Quality & Transparency
Every batch tested. Every result published. No exceptions.
Nicotinamide adenine dinucleotide (NAD+) is a critical small molecule coenzyme found in all living cells, functioning as a central regulator of cellular metabolism and energy homeostasis. As a pyridine nucleotide, NAD+ serves as an essential cofactor in redox reactions, facilitating electron transfer in glycolysis, the citric acid cycle, and oxidative phosphorylation. Beyond its metabolic roles, NAD+ functions as a substrate for several enzyme families including sirtuins, poly(ADP-ribose) polymerases (PARPs), and CD38/CD157 ectoenzymes, positioning it as a key signaling molecule in laboratory investigations of aging, metabolic regulation, DNA repair, and cellular stress responses. In preclinical research, NAD+ has emerged as a focal point for studies examining mitochondrial function, circadian rhythm regulation, and age-related cellular decline. The molecule's involvement in fundamental biochemical processes makes it an invaluable research tool for investigating metabolic pathways, energy sensing mechanisms, and the molecular basis of cellular longevity in controlled experimental settings.
NAD+ is a dinucleotide composed of two nucleotides joined through their phosphate groups, with one nucleotide containing an adenine nucleobase and the other containing nicotinamide. The molecule has a molecular weight of 663.43 g/mol and exists in oxidized (NAD+) and reduced (NADH) forms, enabling its function as an electron carrier in redox reactions. The positively charged pyridinium ring in NAD+ accepts hydride ions (H-) during reduction reactions, converting to NADH, which then donates electrons in subsequent oxidative processes. NAD+ demonstrates pH-dependent stability, with optimal stability at physiological pH ranges. In research applications, the molecule exhibits UV absorbance at 260 nm, facilitating spectrophotometric quantification. The compound's role extends beyond redox biochemistry to serve as a consumed substrate for NAD+-dependent enzymes, where the nicotinamide-glycosidic bond is cleaved, releasing nicotinamide and leaving ADP-ribose moieties that modify target proteins. This dual functionality—as both a recyclable electron carrier and a consumable substrate—underlies its diverse experimental applications in cellular metabolism research.
NAD+ serves as a fundamental research reagent for investigating cellular bioenergetics, metabolic regulation, and molecular mechanisms of aging in laboratory settings. Its involvement in multiple enzymatic pathways makes it an essential tool for dissecting complex cellular processes at the molecular level. Preclinical studies utilize NAD+ to examine mitochondrial function, oxidative stress responses, and metabolic pathway flux in controlled experimental systems.
- Mitochondrial respiration assays utilizing isolated mitochondria or permeabilized cells to assess electron transport chain function and ATP synthesis capacity
- Sirtuin enzyme activity assays examining NAD+-dependent deacetylation reactions and their effects on protein function and gene expression
- PARP activation studies investigating DNA damage responses and poly(ADP-ribosyl)ation signaling cascades in cultured cell lines
- Metabolic flux analysis employing NAD+/NADH ratio measurements to evaluate glycolytic versus oxidative metabolism in various cell types
- Cell culture supplementation experiments examining effects of NAD+ availability on cellular stress resistance, proliferation, and metabolic phenotypes
- Enzyme kinetics studies characterizing NAD+-dependent dehydrogenases and oxidoreductases in purified enzyme preparations
- Circadian rhythm research investigating NAD+ biosynthetic pathway oscillations and their relationship to clock gene expression patterns
NAD+ functions at the nexus of multiple critical cellular pathways, serving both as a coenzyme in energy metabolism and as a substrate for signaling enzymes. In glycolysis and the tricarboxylic acid cycle, NAD+ acts as an electron acceptor, becoming reduced to NADH during substrate oxidation. This NADH subsequently donates electrons to Complex I of the mitochondrial electron transport chain, driving oxidative phosphorylation and ATP synthesis. The NAD+/NADH ratio serves as a key metabolic indicator in research settings, reflecting cellular redox state and energy status. Laboratory studies demonstrate that this ratio influences multiple metabolic enzymes, including pyruvate dehydrogenase and lactate dehydrogenase, thereby regulating the balance between oxidative and glycolytic metabolism in experimental systems.
Beyond redox metabolism, NAD+ serves as a consumed substrate for three major enzyme families. Sirtuins (SIRT1-7 in mammals) utilize NAD+ to catalyze protein deacetylation reactions, producing nicotinamide and acetyl-ADP-ribose as products. In preclinical models, sirtuin activity has been linked to regulation of mitochondrial biogenesis, DNA repair pathways, and stress resistance mechanisms. PARP enzymes consume NAD+ during poly(ADP-ribosyl)ation reactions in response to DNA damage, with excessive PARP activation potentially depleting cellular NAD+ pools in experimental conditions. CD38, a multifunctional ectoenzyme, represents a major NAD+ consumer, converting NAD+ to cyclic ADP-ribose (cADPR) and ADP-ribose, molecules that participate in calcium signaling pathways.
The biosynthesis and salvage pathways of NAD+ have emerged as important research targets. The salvage pathway, utilizing nicotinamide phosphoribosyltransferase (NAMPT) to convert nicotinamide back to NAD+, maintains cellular NAD+ levels under conditions of consumption by NAD+-dependent enzymes. Laboratory investigations indicate that NAD+ biosynthetic capacity influences cellular responses to metabolic stress, oxidative damage, and aging-related decline in experimental models, establishing these pathways as central nodes in metabolic regulation research.
Extensive preclinical research has established NAD+ as a critical determinant of cellular metabolic function and stress resistance in experimental systems. Studies employing rodent models have demonstrated that NAD+ levels decline with age across multiple tissues, correlating with decreased mitochondrial function and altered metabolic homeostasis. In vitro investigations using primary cell cultures and immortalized cell lines have shown that NAD+ availability influences cellular responses to oxidative stress, DNA damage, and nutrient deprivation. Research utilizing NAD+ precursors in mouse models has revealed enhancement of mitochondrial respiratory capacity, upregulation of genes involved in oxidative metabolism, and improved performance in metabolic stress paradigms. Cell culture studies examining NAD+-dependent sirtuin signaling have identified molecular mechanisms linking NAD+ availability to mitochondrial biogenesis through PGC-1α activation and regulation of mitochondrial unfolded protein response pathways.
Experimental work in metabolic disease models has provided insights into NAD+'s role in insulin signaling and glucose homeostasis. Rodent studies employing high-fat diet paradigms have shown that interventions supporting NAD+ biosynthesis can influence hepatic lipid metabolism and insulin sensitivity markers at the molecular level. In vitro hepatocyte and myocyte experiments indicate that NAD+ status affects AMPK activation, lipid oxidation gene expression, and mitochondrial respiratory function. Neurobiological research using cultured neurons and brain slice preparations has examined NAD+'s role in axonal regeneration, synaptic plasticity markers, and neuronal stress resistance mechanisms.
Circadian biology research has revealed oscillations in NAD+ levels and biosynthetic enzyme expression that correlate with core clock gene activity in cell culture and animal models. Studies utilizing genetic manipulation of NAD+ biosynthetic pathways in mice have demonstrated bidirectional relationships between NAD+ metabolism and circadian transcriptional programs. These preclinical observations establish NAD+ as a fundamental research tool for investigating the molecular basis of metabolic regulation, cellular aging processes, and stress response pathways in controlled laboratory environments.
Aureum Peptides supplies NAD+ as a lyophilized powder, ensuring optimal stability during storage and transport for research applications. Each production lot undergoes rigorous analytical verification using high-performance liquid chromatography (HPLC) to confirm purity specifications, typically exceeding 98% purity for research-grade material. Molecular identity is verified through mass spectrometry (MS) analysis, confirming the correct molecular weight and structural integrity of the compound. Endotoxin testing is performed using Limulus Amebocyte Lysate (LAL) assays to ensure suitability for cell culture applications where bacterial contamination must be minimized. All analytical data are compiled in a Certificate of Analysis (COA) available for each lot, providing researchers with complete documentation of product specifications. These quality control measures ensure batch-to-batch consistency, supporting reproducible experimental outcomes in demanding laboratory research protocols requiring well-characterized biochemical reagents.
- 1Cantó C et al., Cell Metabolism, 2012 15(6):838-847. PubMed
- 2Imai S et al., Trends in Pharmacological Sciences, 2014 35(6):313-321. PubMed
- 3Yoshino J et al., Cell Metabolism, 2018 27(3):513-528. PubMed
- 4Verdin E, Science, 2015 350(6265):1208-1213. PubMed
- 5Rajman L et al., Cell Metabolism, 2018 27(3):529-547. PubMed
- 6Gomes AP et al., Cell, 2013 155(7):1624-1638. PubMed
- 7Mouchiroud L et al., Cell, 2013 154(2):430-441. PubMed
- 8Camacho-Pereira J et al., Nature Communications, 2016 7:11416. PubMed
- 9Mori V et al., Cell Reports, 2014 7(6):2042-2055. PubMed
- 10Bai P et al., Trends in Endocrinology and Metabolism, 2015 26(12):699-711. PubMed
- 11Nakahata Y et al., Science, 2009 324(5927):654-657. PubMed
- 12Ramsey KM et al., Science, 2009 324(5927):651-654. PubMed
Advancing Research, One Peptide at a Time
Premium quality. Rigorous testing. Trusted by researchers worldwide.
Third-party testing data will be displayed here once available.
Temperature
Lyophilized: -20°C
Reconstituted: 2-8°C (30 days)
Shelf Life
24 months lyophilized
30 days reconstituted
Handling
Avoid freeze-thaw cycles.
Use bacteriostatic water for reconstitution.
Frequently Asked Questions
Stay at the Forefront
Join our research community. Get early access to new peptides, exclusive member pricing, and curated literature reviews delivered to your inbox.
Welcome to the Circle ✦
Check your inbox for your 10% welcome discount