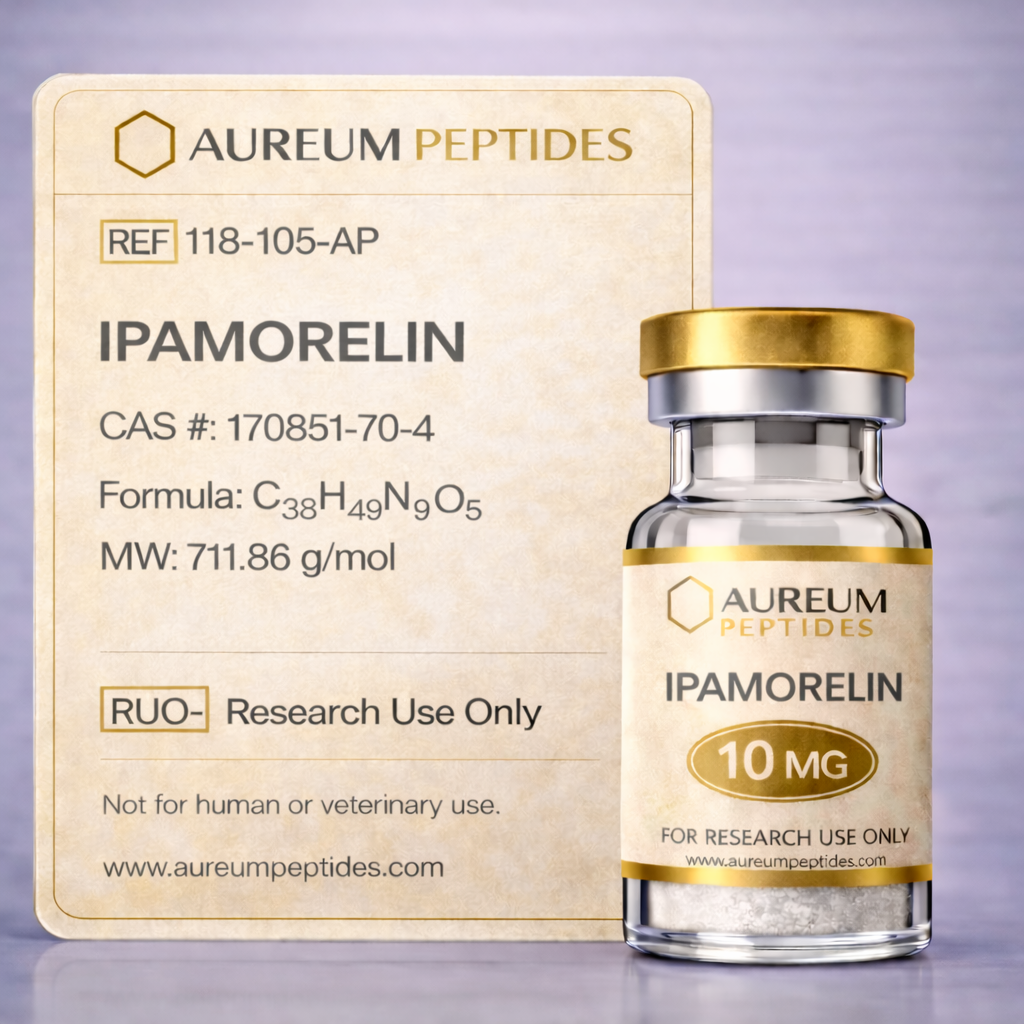

Ipamorelin
NNC 26-0161, NNC-26-0161, Ipamorelin Acetate, Growth Hormone Releasing Peptide-Ipamorelin, GHRP-Ipamorelin

Quality & Transparency
Every batch tested. Every result published. No exceptions.
Ipamorelin is a synthetic pentapeptide classified as a selective growth hormone secretagogue receptor (GHS-R) agonist and a member of the growth hormone-releasing peptide (GHRP) family. Structurally distinct from earlier GHRPs such as GHRP-6 and hexarelin, ipamorelin was developed through rational peptide design to achieve high specificity for growth hormone (GH) release without concurrent stimulation of prolactin or cortisol secretion. This selectivity profile has established ipamorelin as a valuable research tool in neuroendocrine physiology studies. In laboratory research, ipamorelin is employed to investigate the ghrelin receptor system, pulsatile GH secretion mechanisms, and hypothalamic-pituitary axis function. Preclinical studies suggest its utility in models exploring metabolic regulation, bone density modulation, and age-related changes in somatotropic function. Its clean pharmacological profile—characterized by minimal off-target receptor activation—makes it particularly suitable for experimental designs requiring precise manipulation of the GH axis without confounding hormonal cross-reactivity, positioning it as a preferred compound in contemporary growth hormone secretagogue research.
Ipamorelin is a synthetic pentapeptide with the amino acid sequence Aib-His-D-2-Nal-D-Phe-Lys-NH₂, where Aib denotes α-aminoisobutyric acid and D-2-Nal represents D-2-naphthylalanine. This sequence incorporates non-natural amino acids and D-isomers to enhance proteolytic stability and receptor selectivity. The compound functions as a synthetic agonist at the ghrelin receptor (growth hormone secretagogue receptor 1a, GHS-R1a), a G-protein coupled receptor predominantly expressed in the pituitary and hypothalamus. Structurally, the inclusion of bulky hydrophobic residues and the C-terminal amidation contribute to high-affinity receptor binding while minimizing interaction with secondary targets such as adrenocorticotropic hormone (ACTH) or prolactin release pathways. In vitro binding studies demonstrate nanomolar affinity for GHS-R1a with negligible activity at other receptor subtypes. The peptide exhibits good stability in aqueous solutions when lyophilized and reconstituted under controlled pH conditions, though it remains susceptible to enzymatic degradation by peptidases in biological matrices, necessitating appropriate storage and handling protocols in laboratory settings to maintain molecular integrity throughout experimental procedures.
Ipamorelin serves as a pharmacological probe in neuroendocrine research, particularly in investigations of growth hormone secretion dynamics and ghrelin receptor signaling. Its selective activation of GHS-R1a without cortisol or prolactin co-release makes it an ideal tool for dissecting specific components of the hypothalamic-pituitary-somatotropic axis. In preclinical models, ipamorelin is utilized to examine pulsatile GH release patterns, receptor desensitization kinetics, and the interplay between endogenous ghrelin and synthetic secretagogues. Specific experimental applications include:
- Receptor binding assays to characterize GHS-R1a pharmacology and structure-activity relationships
- In vitro pituitary cell culture systems investigating somatotroph calcium signaling and GH exocytosis
- Rodent models examining age-related decline in GH pulse amplitude and frequency
- Studies of metabolic signaling pathways including IGF-1 expression and AMPK activation
- Bone biology research exploring osteoblast differentiation and mineralization markers
- Body composition analysis in laboratory animals assessing lean mass and adipose tissue distribution
- Comparative pharmacology studies evaluating selectivity profiles among GHRP family members
- Hypothalamic explant preparations investigating GHRH neuron activity and arcuate nucleus function
Ipamorelin exerts its primary biological effects through selective agonism of the growth hormone secretagogue receptor type 1a (GHS-R1a), a seven-transmembrane G-protein coupled receptor. Upon binding, the peptide stabilizes an active receptor conformation that couples preferentially to Gαq/11 proteins, initiating phospholipase C (PLC) activation and subsequent generation of inositol 1,4,5-trisphosphate (IP₃) and diacylglycerol (DAG). This signaling cascade mobilizes intracellular calcium stores in anterior pituitary somatotrophs, triggering calcium-dependent exocytosis of growth hormone-containing secretory granules. In vitro electrophysiology studies demonstrate that ipamorelin-induced depolarization occurs through closure of ATP-sensitive potassium channels and opening of voltage-gated calcium channels, producing characteristic burst-firing patterns in somatotroph populations.
At the hypothalamic level, preclinical research indicates that GHS-R1a activation by ipamorelin may modulate growth hormone-releasing hormone (GHRH) neuron activity in the arcuate nucleus, though the peptide's primary site of action appears to be directly at the pituitary gland. Unlike first-generation GHRPs, ipamorelin demonstrates minimal cross-reactivity with receptors mediating ACTH or prolactin release, suggesting conformational selectivity in its receptor activation profile. Downstream of acute GH secretion, laboratory models show time-dependent elevation of hepatic insulin-like growth factor 1 (IGF-1) mRNA expression and circulating IGF-1 protein levels, reflecting activation of the somatotropic axis.
In experimental models, repeated ipamorelin administration has been observed to maintain responsiveness with reduced tachyphylaxis compared to earlier secretagogues, suggesting distinct receptor trafficking or desensitization kinetics. Studies in cell culture systems indicate that ipamorelin-activated GHS-R1a undergoes β-arrestin recruitment and internalization, but with faster receptor recycling rates, potentially accounting for sustained signaling competence across multiple stimulation cycles in research protocols.
Preclinical investigations of ipamorelin have extensively characterized its growth hormone secretagogue properties across multiple experimental systems. In vitro studies using primary rat pituitary cell cultures demonstrate dose-dependent GH release with EC₅₀ values in the nanomolar range, with peak responses occurring 15-30 minutes post-administration. These cell-based assays confirm the peptide's selectivity, showing negligible stimulation of prolactin or ACTH secretion even at concentrations exceeding those required for maximal GH response. Radioligand binding displacement studies reveal high-affinity competition at GHS-R1a with minimal interaction at structurally related receptors, validating the compound's pharmacological specificity.
Rodent model studies have provided substantial data on ipamorelin's in vivo secretagogue activity. Subcutaneous and intravenous administration in rats produces robust, transient elevations in circulating GH with peak levels observed 30-60 minutes post-injection, followed by secondary increases in IGF-1 concentrations over subsequent hours. Importantly, these models demonstrate preservation of physiological GH pulse architecture rather than sustained non-physiological elevation, a characteristic distinguishing ipamorelin from continuous GHRH infusion paradigms. Repeated administration studies in aged rodents show maintained secretagogue efficacy without tolerance development over multi-week protocols, contrasting with desensitization patterns observed with some earlier GHRP compounds.
Body composition analyses in preclinical models have documented increases in lean tissue mass and corresponding decreases in adipose tissue percentage following chronic ipamorelin administration schedules. Bone density measurements using micro-CT imaging in ovariectomized rat models indicate positive effects on trabecular bone volume and cortical thickness, outcomes attributed to GH/IGF-1 axis activation. Molecular analyses of skeletal muscle tissue reveal upregulation of IGF-1 receptor expression and downstream Akt/mTOR pathway phosphorylation status. These observations collectively position ipamorelin as a research tool for investigating somatotropic regulation of anabolic processes in laboratory settings.
Ipamorelin is supplied as a sterile, lyophilized white powder in sealed glass vials, optimized for stability during storage and transport. Each batch undergoes rigorous analytical verification to ensure consistency and purity for research applications. High-performance liquid chromatography (HPLC) analysis confirms peptide purity ≥98%, with detailed chromatographic profiles documenting separation of the target compound from potential synthesis byproducts and degradation fragments. Mass spectrometry (LC-MS/MS) provides definitive molecular weight confirmation and sequence identity verification, ensuring the supplied material matches the expected pentapeptide structure. Endotoxin testing via Limulus Amebocyte Lysate (LAL) assay verifies levels <1.0 EU/mg, meeting standards for cell culture applications. Certificates of Analysis (COA) accompanying each product lot detail these analytical results, supporting experimental reproducibility and compliance with laboratory quality standards. The lyophilized format facilitates controlled reconstitution in appropriate buffers, allowing researchers to prepare working solutions at defined concentrations for their specific experimental protocols.
- 1Raun K et al., Endocrinology, 1998 139(9):3887-3896. PubMed
- 2Gobburu JV et al., Journal of Clinical Pharmacology, 1999 39(1):94-103. PubMed
- 3Svensson J et al., European Journal of Endocrinology, 2000 142(3):270-276. PubMed
- 4Andersson P et al., Growth Hormone & IGF Research, 2001 11(4):200-206. PubMed
- 5Johansen PB et al., European Journal of Endocrinology, 1999 140(4):330-333. PubMed
- 6Grande C et al., Journal of Endocrinology, 2004 181(1):93-103. PubMed
- 7Lall S et al., Journal of Clinical Endocrinology & Metabolism, 2004 89(3):1445-1452. PubMed
- 8Holst B et al., Molecular Endocrinology, 2005 19(9):2400-2411. PubMed
- 9Teichman SL et al., Growth Hormone & IGF Research, 2006 16(5-6):267-278. PubMed
- 10Gevrey JC et al., Endocrinology, 2002 143(11):4439-4442. PubMed
- 11Møller L et al., Growth Hormone & IGF Research, 2015 25(5):219-226. PubMed
- 12Bowers CY, Journal of Pediatric Endocrinology & Metabolism, 2001 14(Suppl 5):1395-1406. PubMed
Advancing Research, One Peptide at a Time
Premium quality. Rigorous testing. Trusted by researchers worldwide.
Third-party testing data will be displayed here once available.
Temperature
Lyophilized: -20°C
Reconstituted: 2-8°C (30 days)
Shelf Life
24 months lyophilized
30 days reconstituted
Handling
Avoid freeze-thaw cycles.
Use bacteriostatic water for reconstitution.
Frequently Asked Questions
Stay at the Forefront
Join our research community. Get early access to new peptides, exclusive member pricing, and curated literature reviews delivered to your inbox.
Welcome to the Circle ✦
Check your inbox for your 10% welcome discount