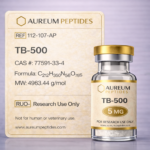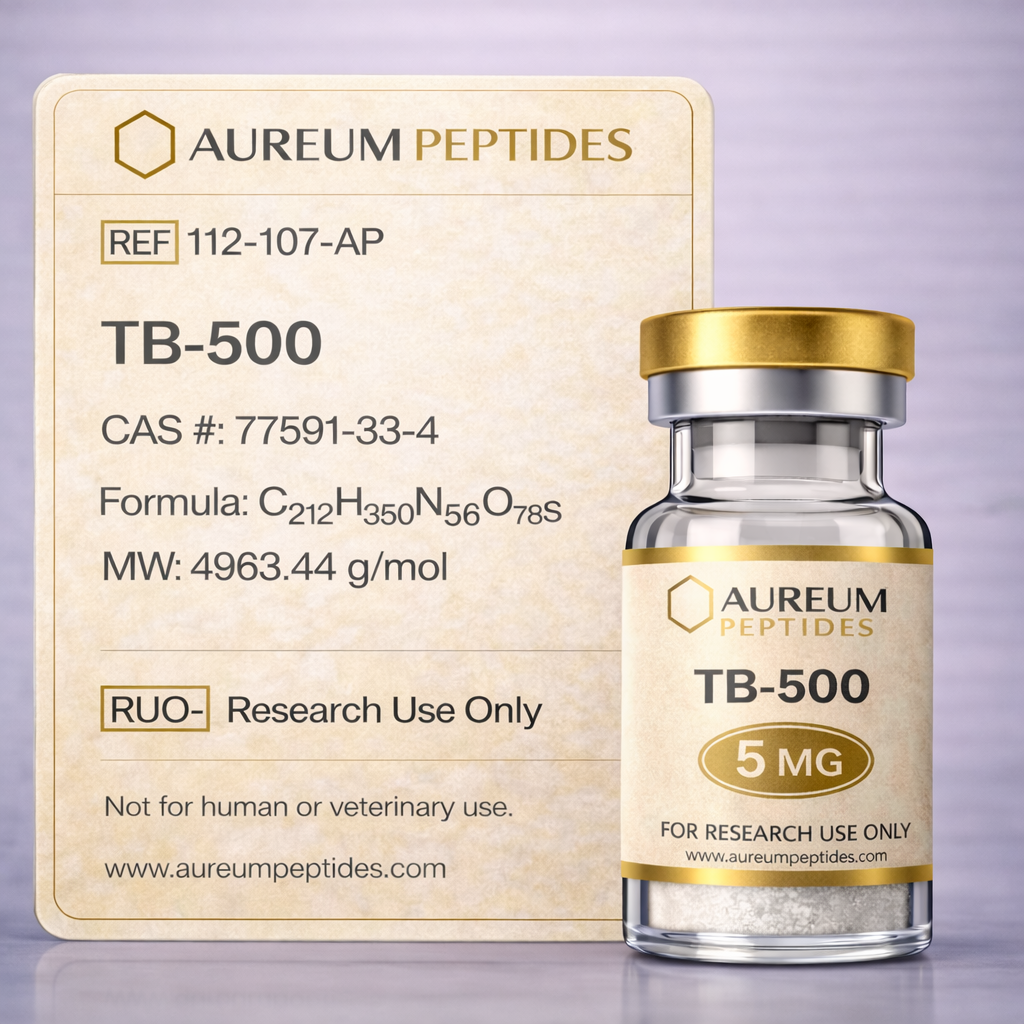

TB-500
Thymosin Beta-4 Fragment, TB4-Fragment, Tβ4 Active Region, 17-23 Region Thymosin Beta-4, TMSB4X Fragment, Thymosin Beta 4 Peptide Fragment, TB 500, TB-4 Fragment
Quality & Transparency
Every batch tested. Every result published. No exceptions.
TB-500 is a synthetic peptide representing a fragment of the naturally occurring protein Thymosin Beta-4, an endogenous peptide present in most mammalian cells. This research-grade compound consists of a 43-amino acid sequence derived from the central active region of Thymosin Beta-4 and has become a focal point in cellular biology and regenerative research models. TB-500 is classified as a bioactive peptide and is supplied exclusively for research use only in laboratory investigations. Primary research interest areas include cellular migration, angiogenic processes in preclinical models, tissue remodeling pathways, and wound healing mechanisms at the molecular level. In vitro and in vivo research has examined TB-500's interactions with actin, a structural protein involved in cellular motility and cytoskeletal dynamics. Investigators in fields such as developmental biology, vascular research, and musculoskeletal research utilize this peptide to explore cell behavior, extracellular matrix remodeling, and molecular signaling cascades associated with tissue repair in experimental systems.
TB-500 is a synthetic peptide comprising 43 amino acids, corresponding to a functional fragment of the 43-amino acid full-length Thymosin Beta-4 protein. This fragment retains the active sequence responsible for actin binding, a critical feature underlying its biochemical activity. The peptide exhibits a molecular weight of approximately 4.9 kDa and possesses an acetylated N-terminus in its native form, though synthetic preparations may vary. Structurally, TB-500 contains multiple charged residues contributing to its solubility profile in aqueous research buffers. The peptide's primary biochemical characteristic is its capacity to sequester globular actin (G-actin), thereby modulating actin polymerization dynamics within cellular systems. This actin-binding property influences cytoskeletal organization and cellular motility in experimental models. TB-500 demonstrates stability when stored as lyophilized powder under appropriate conditions (-20°C or lower), with reconstituted solutions requiring careful handling to maintain peptide integrity. The peptide's hydrophilic nature facilitates dissolution in sterile water or buffered saline solutions commonly employed in laboratory research protocols.
TB-500 serves as a valuable research tool in preclinical investigations examining cellular migration, tissue remodeling, and molecular mechanisms underlying regenerative processes. Laboratory researchers employ this peptide to dissect actin-cytoskeleton interactions and explore cellular responses to injury-mimicking conditions in controlled experimental systems. The compound has been incorporated into diverse in vitro and in vivo models to investigate molecular pathways associated with angiogenesis, extracellular matrix dynamics, and cell survival signaling cascades.
- Cell migration and motility assays in endothelial cell cultures to examine angiogenic mechanisms
- Actin polymerization studies and cytoskeletal dynamics investigations in various cell lines
- In vivo wound healing models in rodents to characterize molecular markers of tissue repair
- Gene expression profiling experiments to identify transcriptional responses to actin-binding peptides
- Vascular development studies in zebrafish and murine models to examine vessel formation pathways
- Inflammatory response characterization in macrophage and immune cell culture systems
- Cardiomyocyte culture experiments investigating cellular survival pathways and stress responses
- Skeletal muscle injury models in mice and rats examining molecular repair mechanisms
TB-500's molecular activity centers on its interaction with monomeric G-actin, where the peptide functions as an actin-sequestering agent. This biochemical interaction influences the equilibrium between globular and filamentous actin (F-actin), thereby modulating cytoskeletal architecture and associated cellular processes. Research observations indicate that by binding G-actin, TB-500 may influence cellular motility, lamellipodia formation, and directional migration in experimental cell culture systems. These actin-dependent processes are fundamental to angiogenesis, wound healing, and tissue remodeling pathways examined in preclinical research contexts.
Beyond actin sequestration, laboratory investigations have explored TB-500's involvement in multiple signaling cascades. Preclinical studies suggest the peptide may modulate the phosphatidylinositol 3-kinase (PI3K)/Akt pathway, a critical signaling cascade regulating cell survival, proliferation, and metabolism. In vitro models indicate potential interactions with integrin-mediated signaling pathways, which coordinate cell-extracellular matrix interactions and mechanotransduction processes. Research has also examined TB-500's influence on vascular endothelial growth factor (VEGF) expression and related pro-angiogenic signaling networks in experimental systems.
Gene expression analyses in preclinical models have identified TB-500-associated changes in transcriptional programs related to inflammation modulation, including alterations in cytokine and chemokine expression profiles. Laboratory observations suggest the peptide may influence NF-κB signaling pathway components, though the precise molecular mechanisms remain under investigation. Additionally, research has examined TB-500's potential role in matrix metalloproteinase (MMP) regulation and extracellular matrix remodeling processes, which are central to tissue repair mechanisms studied in regenerative research models. These pathway-level observations provide investigators with molecular targets for mechanistic exploration in controlled research settings.
Preclinical investigations utilizing TB-500 have generated extensive molecular-level observations across diverse experimental systems. In rodent wound healing models, researchers have documented accelerated closure rates accompanied by enhanced collagen deposition and altered inflammatory cell infiltration patterns when compared to control groups. At the molecular level, these models have revealed increased expression of genes associated with cell migration, angiogenesis, and extracellular matrix remodeling. In vitro endothelial cell studies have demonstrated enhanced migration and tube formation capabilities in TB-500-treated cultures, with accompanying upregulation of VEGF and other pro-angiogenic factors, suggesting involvement in vascular development pathways. Cardiomyocyte culture experiments have shown reduced apoptotic markers and enhanced survival signaling following experimentally induced stress conditions in TB-500-exposed cells.
Musculoskeletal research utilizing murine models of muscle injury has characterized TB-500's association with molecular markers of regeneration, including satellite cell activation markers and myogenic regulatory factor expression profiles. These preclinical observations have been accompanied by histological documentation of enhanced myofiber organization and reduced fibrotic tissue deposition in experimental injury sites. Rodent models examining tendon and ligament injury have similarly revealed altered biomolecular profiles in TB-500-exposed tissues, including modified collagen organization patterns and inflammatory mediator expression.
Neurological research models have explored TB-500's effects in experimental systems mimicking ischemic conditions. In vitro neuronal cultures exposed to oxygen-glucose deprivation have exhibited reduced cell death markers in the presence of TB-500, while rodent stroke models have documented reduced infarct volumes and altered expression of neurotrophic factors and inflammatory markers. These diverse preclinical observations, spanning cardiovascular, musculoskeletal, dermatological, and neurological research domains, underscore TB-500's utility as a research tool for investigating actin-dependent cellular processes and molecular pathways associated with tissue homeostasis and repair mechanisms in controlled laboratory settings.
TB-500 is supplied as a sterile, lyophilized powder, a form optimized for stability during storage and transport in research settings. This presentation facilitates precise reconstitution according to experimental protocol requirements. Each production batch undergoes comprehensive analytical verification to ensure consistency for laboratory workflows. High-performance liquid chromatography (HPLC) is employed to quantify peptide purity, with specifications typically exceeding 98%, thereby minimizing interfering substances in experimental systems. Mass spectrometry analysis confirms molecular identity and structural integrity, verifying the correct amino acid sequence and expected molecular weight. Endotoxin testing using Limulus Amebocyte Lysate (LAL) assays ensures bacterial pyrogen levels remain below specified thresholds, critical for cell culture applications and in vivo research models. Certificates of Analysis (COA) are available for each lot, providing researchers with batch-specific analytical data including purity profiles, mass spectral data, and endotoxin levels, supporting reproducibility and quality documentation requirements in laboratory research environments.
- 1Goldstein AL et al., Ann N Y Acad Sci, 2007 1112:1-13. PubMed
- 2Sosne G et al., Ocul Surf, 2010 8(1):5-17. PubMed
- 3Philp D et al., J Cell Sci, 2003 116(Pt 19):3947-3955. PubMed
- 4Bock-Marquette I et al., Nature, 2004 432(7016):466-472. PubMed
- 5Badamchian M et al., Ann N Y Acad Sci, 2007 1112:154-160. PubMed
- 6Smart N et al., Nature, 2007 445(7124):177-182. PubMed
- 7Crockford D et al., Expert Opin Biol Ther, 2010 10(1):19-25. PubMed
- 8Smalheiser NR et al., Brain Res, 1996 728(2):269-273. PubMed
- 9Huff T et al., Int J Biochem Cell Biol, 2001 33(3):205-220. PubMed
- 10Saidi H et al., Ann Thorac Surg, 2003 75(6):1787-1793. PubMed
- 11Malinda KM et al., J Cell Physiol, 1997 173(1):74-81. PubMed
- 12Guarnera E et al., J Biol Chem, 2009 284(40):27179-27188. PubMed
Advancing Research, One Peptide at a Time
Premium quality. Rigorous testing. Trusted by researchers worldwide.
Third-party testing data will be displayed here once available.
Temperature
Lyophilized: -20°C
Reconstituted: 2-8°C (30 days)
Shelf Life
24 months lyophilized
30 days reconstituted
Handling
Avoid freeze-thaw cycles.
Use bacteriostatic water for reconstitution.
Frequently Asked Questions
Stay at the Forefront
Join our research community. Get early access to new peptides, exclusive member pricing, and curated literature reviews delivered to your inbox.
Welcome to the Circle ✦
Check your inbox for your 10% welcome discount